The Health and Economic Implications of Antibiotic Overuse and Resistance
• Opinion
“Regarding human health, the spread of antibiotic resistant bacteria is one of the most critical public health issues worldwide.”[1]
Antimicrobials are drugs that destroy disease-causing microbes, such as bacteria, viruses, parasites and fungi. The most familiar and important antimicrobials are antibiotics - arguably humanity’s greatest medical invention and often referred to as miracle drugs. Since penicillin was first used to treat humans in 1941, they have saved hundreds of millions of lives, drastically changed modern medicine, and extended the average human lifespan by 23 years.[2] However, our overuse and misuse of antimicrobials puts these life‑saving drugs at risk.
The Growing Threat of Antimicrobial Resistance
In recent decades, antibiotic misuse has resulted in the rapid rise of antimicrobial resistance (”AMR”), with some infections now effectively untreatable.[3] Because AMR emerges at the intersection of human, animal, and environmental health, a framework known as the One Health concept, it has been called “the quintessential One Health problem,” and directly resulted in 1.27 million deaths in 2019.[4]
“A post-antibiotic era means, in effect, an end to modern medicine as we know it. Things as common as strep throat or a child’s scratched knee could once again kill”, explains Margaret Chan, former director-general of the World Health Organization.[5]
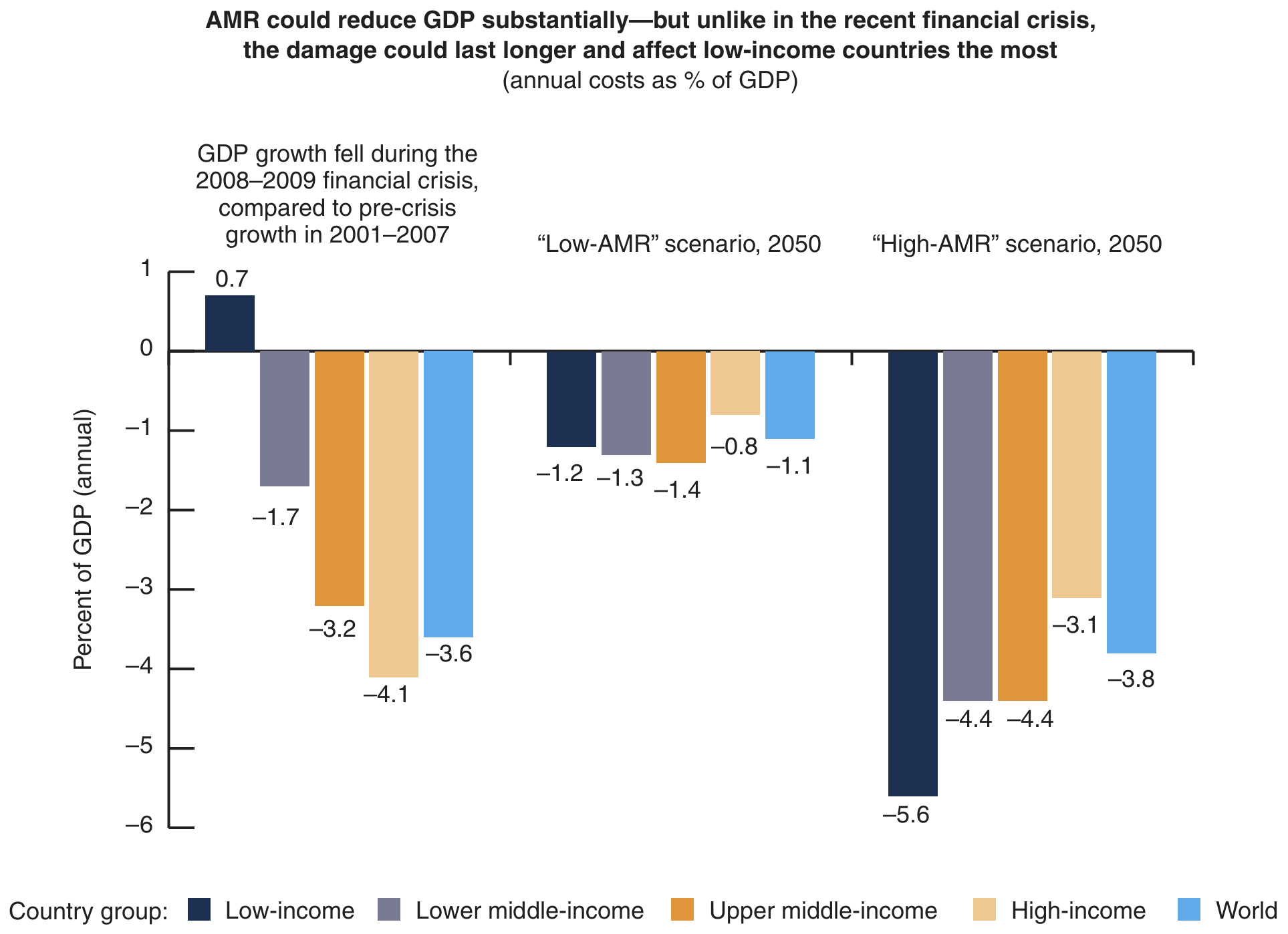
The consequences of AMR extend beyond healthcare, they ripple through economies too. The World Bank estimates that AMR will likely cause global GDP to fall between 1.1% and 3.8% by 2050, with an annual global shortfall between $1 trillion and $3.4 trillion by 2030.[6]
Figure 1: Substantial and protracted shortfalls in global economic output
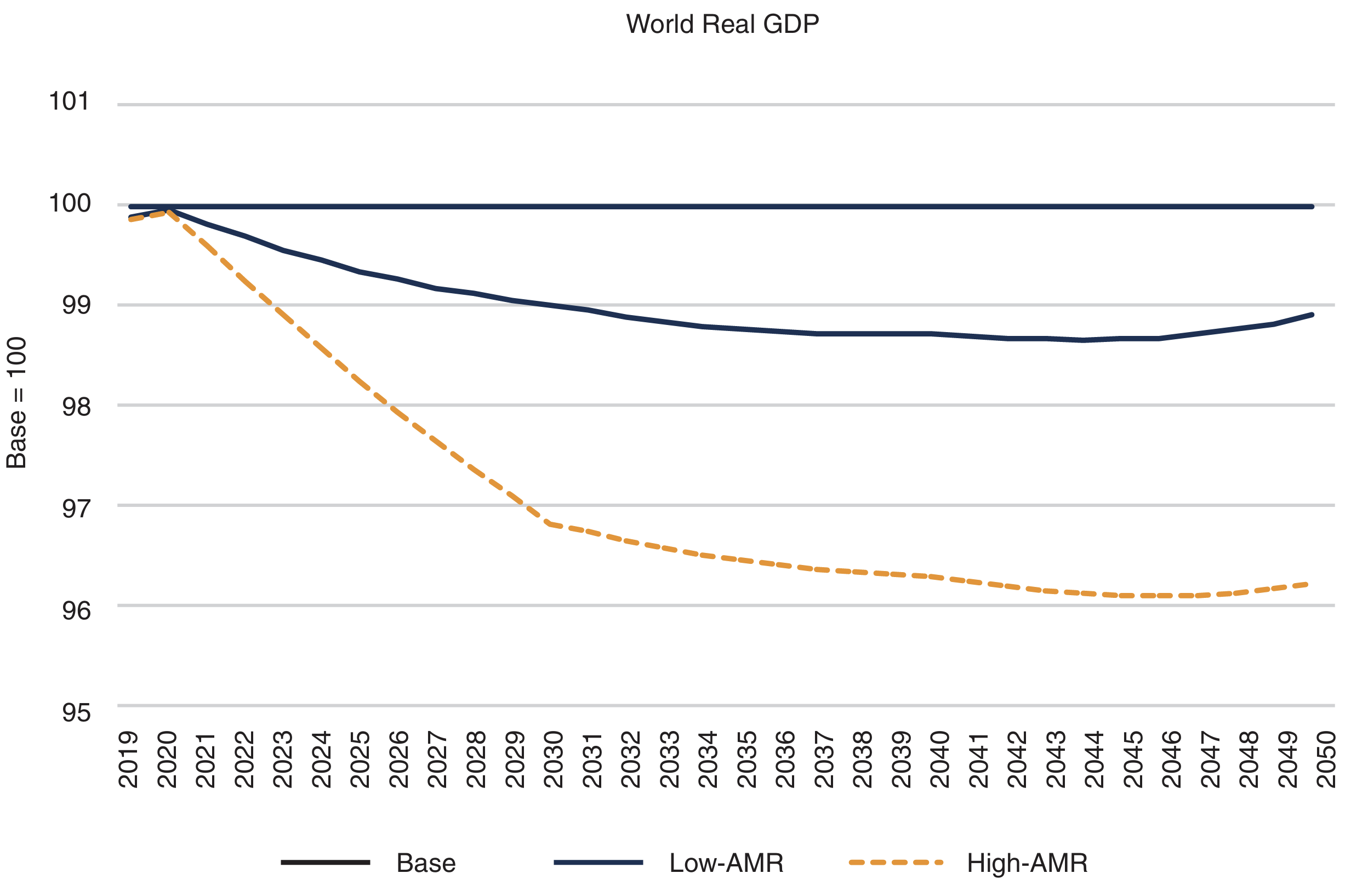
During much of the period through 2050, the annual reduction in GDP caused by AMR could be as large as the losses provoked by the 2008-2009 global financial crisis, with low-income countries experiencing larger drops in economic growth due to higher infectious disease prevalence and greater dependence on labour incomes.
Figure 2: Economic costs of AMR may be as severe as during the financial crisis
The costs of some of the measures that will be required to contain AMR vary widely across countries. The below figure shows just one example: the cost differentials in treating tuberculosis (TB) with and without AMR, when bacteria are multidrug-resistant (MDR) and extensively drug-resistant (XDR). Note that treatment costs are much higher in high-income countries than in low-income countries because the costs of medical personnel and supplies is much higher. Thus, the World Bank Group concludes it would cost 80 times more to treat one TB patient in the US than in India.[6] This provides a powerful rationale for all countries to explore how best to curve AMR. To understand why resistance is accelerating so quickly, we need to understand where majority of antibiotics are actually used.
Figure 3: AMR makes TB far costlier to treat

Our Food System is a Key Driver of Antibiotic Overuse and Resistance
What’s often left unsaid about antimicrobial resistance is who the real takers of these drugs are. We tend to think only of humans, but a far larger share goes to farm animals.
Researchers have estimated that in the 2010s, roughly 70% of the world’s antibiotics went to livestock.[7] Worldwide, in 2010, livestock consumed at least 63,200 tons of antibiotics, exceeding total human consumption.[6] Poor hygienic conditions and the extreme density of animals on many farms create ideal environments for pathogens to emerge, mutate rapidly and spread, significantly increasing disease risks for both animals and humans.
The overuse of antibiotics in farm animals accelerates the emergence of drug‑resistant bacteria, increasing the risk that these pathogens will ultimately infect humans. Bacteria such as E. coli and Salmonella enterica, along with the resistance genes they acquire, can spread rapidly. Once resistant pathogens take hold in animals or people, they can move through human‑to‑human, animal‑to‑human, and animal‑to‑animal routes. They can even spread resistance horizontally between microbes, including across different species.
As the COVID‑19 pandemic demonstrated, infectious threats cannot be contained geographically. The same is true for antibiotic resistant pathogens, which spread across regions.
Figure 4: Antibiotic use in livestock compared to proposed thresholds*
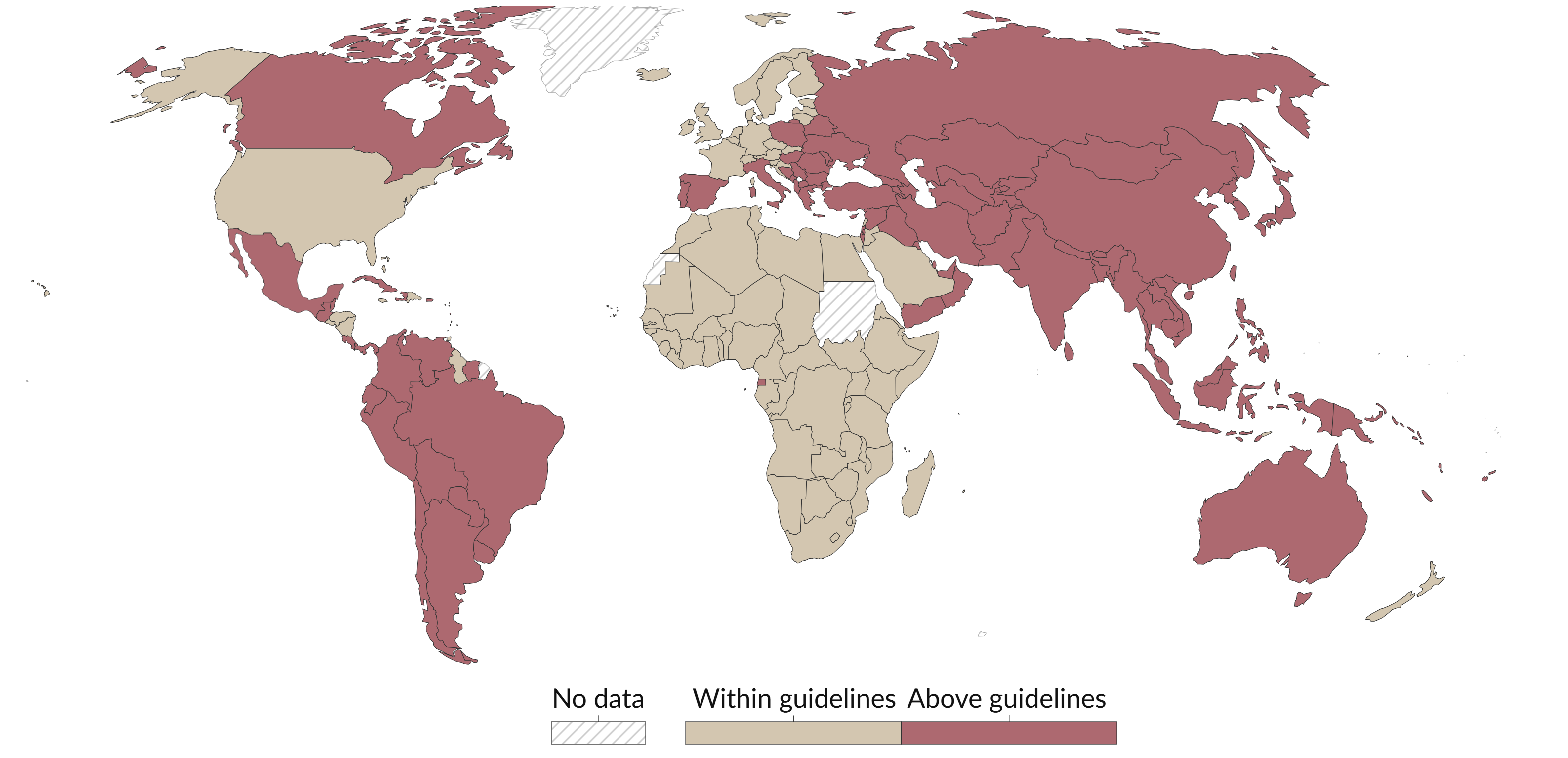
*Note: Researchers working on antimicrobial resistance have proposed a threshold of 50mg per PCU, which is shown as the threshold here.[8]
Food Tech as a Solution
“Producing meat through alternative methods, such as cultivated meat, offers an opportunity to decouple antibiotics from meat.”[9]
Food tech solutions that decouple meat production from live animals present an opportunity to reduce antibiotic use dramatically. Cell culture, precision fermentation, and plant-based technologies are some of the ways we can produce the foods we love without animals.
In cultivated meat, the equivalent of an animal is a bioreactor, where meat is grown from animal cells without the need to raise or slaughter livestock. As you’d expect, conditions in these “farms” are far more sterile and controlled, meaning there’s no need for antibiotics to prevent disease in the “herd”.[9]
For example, if a “farm” like Upside Foods (a Synthesis cultivated meat portfolio company) can supply a city’s chicken demand, it avoids the need to raise millions of broilers dosed with the antibiotic tetracycline, thereby avoiding the tons of tetracycline-laden waste those chickens would have produced, which may have finally entered the city’s sewage systems, water sources and soil.[10, 11] All such waste can promote AMR in microbes in the environment.
Similarly, plant-based meat companies can make highly realistic plant-based meat entirely without the use of antibiotics. Redefine Meat (another Synthesis portfolio company) produces highly realistic and delicious plant-based meats.
Across food technologies, from plant-based to cultivated meat, the common thread is clear: producing food without animals prevents harmful externalities at their source. These food tech innovations are our best bet to meet the growing demand for animal protein without the huge amount of antibiotics that conventional animal agriculture depends on.
Closing Thoughts
By reducing livestock antibiotic use through scaling food‑tech innovations, we can build a food system that protects human health while strengthening the resilience of our protein supply. If adopted at scale, alternative proteins could meaningfully lower antibiotic use globally, helping slow the spread of antimicrobial resistance at its source. By investing in new ways to produce the foods people rely on, we can help preserve the effectiveness of essential medicines and strengthen global public health.
Sources
- Alkorta, I. & Garbisu, C. 2025. Expanding the focus of the One Health concept: links between the Earth-system processes of the planetary boundaries framework and antibiotic resistance. Reviews on Environmental Health, 40(1), 159-173. https://doi.org/10.1515/reveh-2024-0013
- Matthew I Hutchings, Andrew W Truman, Barrie Wilkinson. 2019. Antibiotics: past, present and future. Current Opinion in Microbiology, 51, 72-80. https://doi.org/10.1016/j.mib.2019.10.008
- John F. Prescott. 2014. The resistance tsunami, antimicrobial stewardship, and the golden age of microbiology. Veterinary Microbiology, 171(3-4), 273-278. https://doi.org/10.1016/j.vetmic.2014.02.035
- World Health Organization. 2023. Antimicrobial resistance. Available online
- The Washington Post. 2012. Resistance to antibiotics is becoming a crisis. Available online
- Jonas, Olga B.; Irwin, Alec; Berthe, Franck Cesar Jean; Le Gall, Francois G.; Marquez, Patricio Vicente. Drug-resistant infections : a threat to our economic future (Vol. 2 of 2) : final report (English). HNP/Agriculture Global Antimicrobial Resistance Initiative Washington, D.C. : World Bank Group. http://documents.worldbank.org/curated/en/323311493396993758
- Hannah Ritchie and Fiona Spooner. 2024. Large amounts of antibiotics are used in livestock, but several countries have shown this doesn’t have to be the case. Published online at OurWorldinData.org. Retrieved from: 'https://archive.ourworldindata.org/20251209-133038/antibiotics-livestock.html
- Van Boeckel, T. P., Brower, C., Gilbert, M., Grenfell, B. T., Levin, S. A., Robinson, T. P., ... & Laxminarayan, R. 2015. Global trends in antimicrobial use in food animals. Proceedings of the National Academy of Sciences
- McNamara, E., Bomkamp, C. 2022. Cultivated meat as a tool for fighting antimicrobial resistance. Nat Food. 3, 791–794. https://doi.org/10.1038/s43016-022-00602-y
- Youcef Mehdi, Marie-Pierre Létourneau-Montminy, Marie-Lou Gaucher, Younes Chorfi, Gayatri Suresh, Tarek Rouissi, Satinder Kaur Brar, Caroline Côté, Antonio Avalos Ramirez, Stéphane Godbout. 2018. Use of antibiotics in broiler production: Global impacts and alternatives. Animal Nutrition, 4(2), 170-178. https://doi.org/10.1016/j.aninu.2018.03.002
- Changning Li, Mukesh Kumar Awasthi, Jie Liu, Tuo Yao. 2025. Veterinary tetracycline residues: Environmental occurrence, ecotoxicity, and degradation mechanism. Environmental Research, 266, 120417. https://doi.org/10.1016/j.envres.2024.120417